Request More Information
Monoclonal Antibodies and Biosimilars
Monoclonal antibodies (mAbs) have emerged as revolutionary therapeutic agents for an array of human diseases. In addition to their large mass and complex structure, mAbs are varied in their origin, makeup, effector function and delivery, and therefore require thorough formulation development, including characterization, quantitation and preservation.
More and more special designed Antibody drug conjugates (ADCs) are under development as targeted therapy for treating cancer. ADCs combine the targeting capabilities of monoclonal antibodies with the cancer-killing ability of cytotoxic drugs, allowing new “proximity” based dosing strategies.
As therapeutic development moves from IgG-based antibodies to technologies like bispecific antibodies, peptibodies, and fusion proteins, we are leading innovation in analytical techniques. nSMOL (nano-surface and molecular orientation limited proteolysis) is Shimadzu’s novel approach that enables selective proteolysis of the Fab region of monoclonal antibodies. The nSMOL Antibody BA Kit is a ready-to-use reagent kit for collecting monoclonal antibodies from blood or other biological samples using immunoglobulin collection resin, and then performing selective proteolysis of the Fab region of these antibodies via trypsin-immobilized nanoparticles. Variable region-derived peptides produced by limited proteolysis can then be quantified via MRM measurements utilizing a high-performance liquid chromatograph mass spectrometer.
As first and second generation antibody therapeutics migrate off patent, there is a need to provide the same robust analysis to biosimilar therapeutics. The need to characterize not only the sequence of expressed mAbs, but the various post-translational modifications (PTMs) that can occur is key to proving bioequivalence for regulatory filings. Solutions for N-linked glycan analysis, charge variant analysis, and aggregation state are all available from your partners at Shimadzu.
Analytical Solutions from Cell Culture to Biologics Development and Quality Control
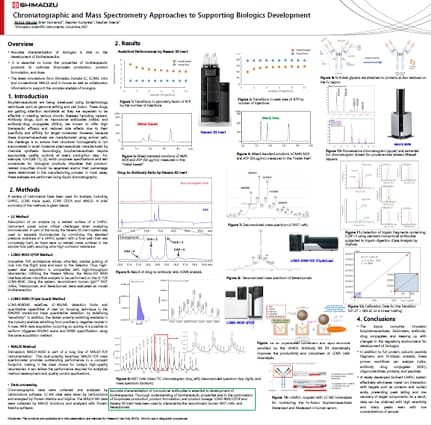
In this study, a multi-instrument platform was used to observe the process of cell culture production to the development of biologics. A CHO cell line was chosen for the study of culture solutions that were sampled every 24 hours and supernatants obtained by centrifugation. An automatic pretreatment with the Shimadzu C2MAP-2030 system was used for removing proteins from culture supernatants by precipitation with organic solvent, suction filtration, and automated transfer of samples for LC-MS/MS analysis (TripleQuad mass spectrometry). In a 20-minute method cycle, the system analyzed 125 metabolites with a wide range of chemical properties simultaneously using high-speed scanning and positive/negative ionization switching. During cell culture, an atomic absorption spectrometry (AAS) system was used to quantify element concentrations based on the absorption of specific light wavelengths. The high concentrations of Mg and Zn were measured by flame AA and the trace elements (Cu, Mn, Co, and Fe) were determined by graphite furnace AA (GFAA). Full characterization of product monoclonal antibodies (mAbs) was undertaken by LCMS, MALDI and UHPLC. Peptide mapping, peptide sequencing, glycosylation, and oxidation of target mAbs analyzed by QTOF, triple quadrupole, and MALDI mass spectrometry was performed to characterize and control biologics production. Additionally, n-glycans were analyzed using UHPLC with fluorescence detection after labeling with pyridylamino (PA)-glycan and 2-aminobenzamide (2-AB)- glycan.
Chromatographic and Mass Spectrometry Approaches to Supporting Biologics Development
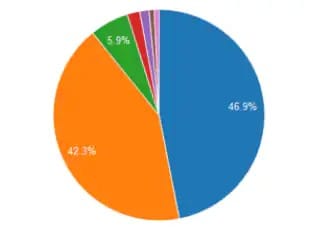
Monoclonal antibodies (mAbs) are a class of biotherapeutics that are rapidly gaining traction. The higher-order structures of mAbs play a crucial role in determining their efficacy and safety. It is crucial that mAbs are accurately characterized before they can become biotherapeutics. A better understanding of mAbs helps craft better bioprocesses, formulas, and dosages. In this study, a recombinant human IgG NIST mAb reference standard was analyzed using high resolution quadrupole time of flight liquid chromatography-mass spectrometry (QTOF-LCMS). An innovative proteolysis method called nano-surface and molecular-orientation limited proteolysis (nSMOL) selectively cleaves Fab regions of mAbs. Regardless of the type of antibodies scientists use, nSMOL can be used to develop analytical methodologies. Triple quadrupole liquid chromatography-mass spectrometry (TQ-LCMS) permits multiple reaction monitoring (MRM) to determine the level of fragmentation in peptides derived from Fab. Furthermore, a protocol for selecting pharmacokinetically suitable signature peptides was developed using nSMOL and TQ-LCMS. Additionally, consistency of glycosylation during process validation is a critical quality attribute (CQA) as N-linked glycans can impact the stability, solubility, and recognition of glycoproteins by glycan-binding proteins. Proteins are attached to N-glycans by covalent bonds formed by N-glycosidic bonds forming at the asparagine (Asn) residues. Monoclonal antibodies and biosimilars can cause such an event at the crystallizable fragment (Fc). In this study, we measured the released glycans from mAbs using QTOF-LCMS. A variety of methodologies are available for the characterization of mAbs and related biologics. Any kind of analysis can be performed based on our tailored workflows, including whole protein analysis, subunit, and peptide fragment analysis, and released N-Glycan analysis.
Featured LC-MS Applications
Monoclonal Antibody Workflows on the LCMS-9030
The Shimadzu Q-TOF LCMS-9030 plus Protein Metrics software offers a complete workflow solution for monoclonal antibody characterization. Customizable workflows can be created for a type of analysis including intact data, subunit, peptide fragments and deglycosylation analysis.
N-Linked Glycan Analysis of Monoclonal Antibodies, Biosimilars, and Glycoproteins with High Resolution Mass Spectrometry and Fluorescence Detection
The Shimadzu LCMS-9030 has shown excellent mass accuracy for N-linked glycans on several proteins. The Restek Raptor Polar X, an innovative hybrid ion-exchange/HILIC column, is an ideal column for glycans as reflected by the baseline separation of the structurally similar N-glycans. Protein Metrics Glycan Workflow offers additional workflows to other published methods.
LCMS Bioanalysis of Antibody Drugs Using Fab-selective Proteolysis "nSMOL Method"
This report describes a selection protocol of signature peptides suitable for pharmacokinetic studies.
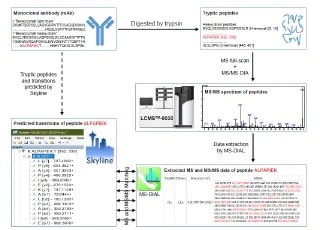
In-depth Peptide Mapping of Monoclonal Antibody (mAb) by A de novo Peptide Sequencing Method on Q-TOF Mass Spectrometer with Data-Independent Acquistion
In this report, we demonstrated an integrated MS full-scan and MS/MS Diag approach on Shimadzu LCMS-9030 (Q-TOF) mass spectrometer for de novo peptide sequencing of mAb.
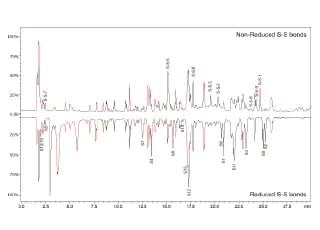
Disulfide Bond Characterization of Monoclonal Antibody (mAb) using Q-TOF Mass Spectrometer
A straightforward LCMS-based method for accurate disulfide bond peptide characterization of mAb biosimilar was established on LCMS-9030 (Q-TOF).The MS/MS spectra with fragmentation data provide high confidence results on sequencing analysis. The demonstrated performance for bevacizumab biosimilar in detection and de novo sequencing of non-reduced and reduced disulfide bonds signifies its practicability for the structural characterization of mAb biosimilars.
Featured MALDI Applications
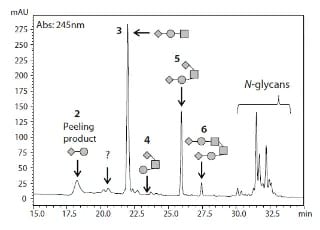
A Study on a Method for Evaluating Glycans in Biopharmaceuticals
In this article, we report the results of studying a method for releasing O-glycans chemically in which the peeling reaction is suppressed, based on a PMP labeling method.
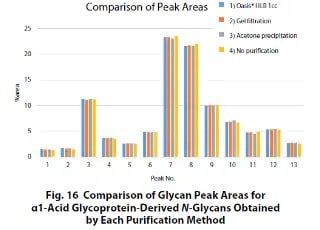
A Study on a Method for Evaluating Glycans in Biopharmaceuticals - Part 2
This article introduces the results of comparing some of the pretreatment methods widely used in N-glycan analysis and investigating how they affect the analysis results.
Characterization of Glycan Binding Site of O-Linked Glycopeptides Using MALDI-7090 High Resolution MALDI-TOF MS
In this article, we present a method for determining the O-linked glycan binding site that uses partial digestion with multiple enzymes and the MALDI-7090 high-resolution MALDI-TOF MS.
Simplified Mass Measurement of Chemically-Modified Antibodies: Determination of the Presence of the Number of Modifications Using a Linear Benchtop MALDI-TOF MS
This article introduces an example of analyzing the pseudo ADC, which was created by artificially binding low-molecular compounds to a standard research antibody, using a benchtop MALDI-TOF MS.
Affinity Purification of IdeZ digest for Glycosylation Profile of Immunoglobulins using a linear benchtop MALDI-TOFMS
Glycosylation on protein plays wide-range vital roles in biological processes from stabilization of protein conformation to expression of binding specificity. A characterization of the N-/O-linked glycan is quite significant, especially in development of biopharmaceuticals. To date, whereas intensive efforts were conducted to characterize glycans precisely with high-end mass spectrometers, conventional instruments without time consuming preparation have been anticipated for batch analysis in screening or QA/QC.
Analysis of Modification Site of Chemically Modified Antibody Using MALDI-Mini-1 Compact MALDI DIT Mass Spectrometer
Antibody drug conjugates (ADC) are a new class of anti-cancer drugs in which an antibody is bound to a cytotoxic drug. Because they combine the high substrate specificity of the antibody and the effect of a low-molecular drug, ADC are expected to be more effective anti-cancer drugs than the conventional low-molecular drugs. When a different compound is bound artificially to a protein, the binding degree of that compound and its binding site become one of the critical quality properties. A pseudo ADC was created by artificially binding a low-molecular compound to a standard research antibody, and was then analyzed using a MALDImini-1.
Analysis of N-Linked Glycan using MALDI-Mini-1 Compact MALDI Digital Ion Trap Mass Spectrometer: Structural Analysis and Identification of Sialyl Linkage Isomers
Focusing on a blood serum-derived N-linked glycan, this article introduces an example in which sialic acid residue was stabilized by using the sialic acid linkage specific alkylamidation (SALSA) method and detection and analysis were performed with a Shimadzu MALDImini-1 compact MALDI digital ion trap (MALDI-DIT) mass spectrometer.
Glycosylation Profile of IgGs Using a Linear Benchtop MALDI-TOFMS and Affinity Purification of Fc
Purification of enzymatically fragmented IgGs by affinity-beads enables batch analysis for the glycosylation using a bench-top MALDI-TOFMS, MALDI-8020. The MS resolution of MALDI-8020 is sufficient to recognize three Fc that mainly differ in glycosylation. A statistical analysis by eMSTAT Solution enables a classification of three glycosylated Fc smoothly and quickly, which could be applicable to QA/QC.
Additional Applications
Analyses of Antibody Drugs Using Ultra High Performance Liquid Chromatography
This article introduces analyses of mAbs and ADCs for quality control using an inert UHPLC system “Nexera XS inert” that is extremely resistant to mobile phases containing high salt concentrations. The type of impurities and their amount are different depending on the type of antibody. There fore, it is note worthy that analytical conditions should be optimized for each sample. Thus, the optimization techniques will also be described in this article.
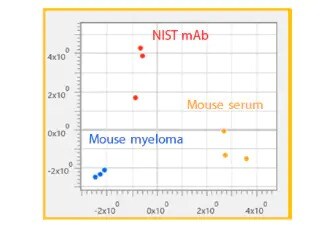
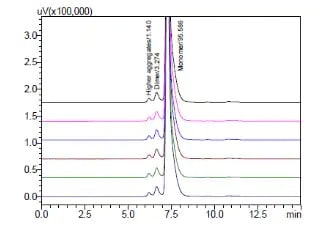
Analysis of protein drugs aggregation Using Size Exclusion Chromatography
This article describes an aggregate analysis using a Shimadzu “Shim-pack™ BioDiol”, size exclusion chromatography column with “Nexera XS inert”, an ultra high performance liquid chromatograph. This chromatograph has high salt tolerance and metal-free flow path and allows to use highly salted mobile phases and prevents sample adsorption.
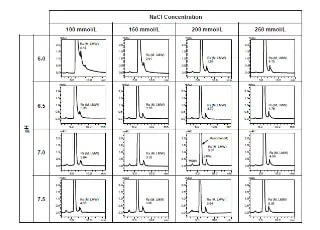
Method Optimization for the Analysis of Monoclonal Antibodies by Size-Exclusion Chromatography
Antibody drugs using monoclonal antibodies pose concerns over aggregates formed during production and storage and their impact on safety and efficacy. During monoclonal antibody production, aggregates formation is monitored, and size-exclusion chromatography is one of the most widely used techniques. However, size-exclusion chromatography is performed at relatively low flow rates and requires long analysis times. Analyses of monoclonal antibody drugs must also take into account interaction between the monoclonal antibodies and column packing materials. This Technical Report provides an example of using a column packed with small particle material to optimize an analytical method for analysis of monoclonal antibody aggregates. This article investigates the effect of mobile phase salt concentration, flow rate, and pH on chromatographic separation and peak shape. Moreover we describe an example of method optimization by using a dedicated software for improving separation, sensitivity and reducing analytical time.
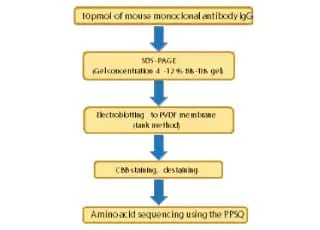
N-Terminal Amino Acid Sequencing of IgG Antibodies
This article introduces an example of amino acid sequencing of mouse antibody IgG using the PPSQ-51A/53A Protein Sequencer isocratic system as an instance of N-terminal amino acid sequencing of biomedicines.
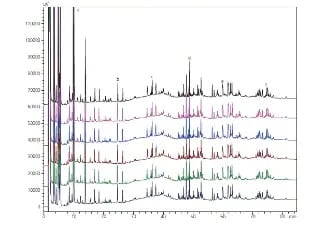
Peptide Mapping of Antibody Drugs by Nexera-i
Peptide mapping by HPLC is one of the important quality assurance tests used for verifying the primary structure of antibody drugs. Typically, following enzymatic digestion of the antibodies, separation is conducted using a traditional reversed phase column. Due to the large number of peaks that require separation, the use of small-particle columns and core shell columns for peptide analysis has spread in recent years. In order to compare elution profiles for identity and mutation confirmation, a highly repeatable system is required. Here, the Nexera-i is used in the analysis of IgG (human immunoglobulin G) tryptic digest.
Analysis of mAb Aggregates by Nexera Bio UHPLC with a Shim-pack Bio Diol (Size Exclusion) Column
The production of mAb biosimilar is challenging, because various variations may occur in upstream and downstream processing(DSP). Protein aggregation is one of such variations ─ a biological phenomenon in which mAb accumulate and clump together. Aggregation is a critical quality attribute (CQAs), as the aggregates not only reduce the efficacy of mAb biosimilar drugs, but also can stimulate immunogenic responses, leading to various adverse events in treatment.
Optimizing Aggregate Analysis - Pore Size
In SEC,smaller molecules enter the pores of the particle and proceed slowly along the axis direction of the column while molecules larger than the pore size are excluded from pores and elute at first from the column.In this application, we demonstrate the effect of pore size in SEC analysis.
Optimizing Aggregate Analysis - Mobile Phase
Despite being a simple assay, mobile phase condition is often needed to be optimized to improve peak shape and resolution of proteins.In this study, we describe analysis of trastuzumab, an anti-HER2 mAb using Shim-pack Bio Diol column on Nexera Bio UHPLC.The effect of mobile phase composition (ionic strength and pH) on chromatographic separation of trastuzumab is presented in this application news.
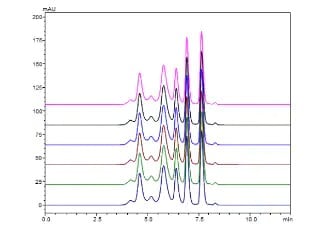
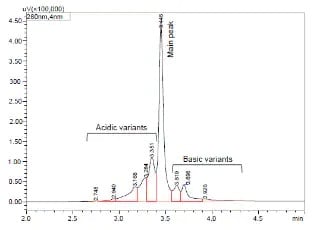
Charge Variant analysis of mAb Biosimilars by Nexera UHPLC with a Shim-pack Bio IEX Column
In this study, we describe both salt-gradient and pH-gradient methods for separating the charge variants of bevacizumab biosimilar using Shimadzu Nexera UHPLC and a Shim-pack Bio IEX column (4.6 mm x100 mm, 3 μm).
Exploring Factors that Influence Charge Variant Analysis of mAbs with Shim-pack Bio IEX Columns using Salt Gradient Elution
In this study, we describe a charge variant analysis of Trastuzumab using salt gradient with Shimadzu strong cation-exchange column, Shim-pack Bio IEX SP-NP. The separation efficiency under different gradient slopes, gradient times and flow rates are discussed.
News / Events
-
AAPS Pharm Sci 360 2024
October 21-23
Salt Palace Convention Center
Salt Lake City, Utah
Booth #2107
-
ASMS (American Society for Mass Spectrometry) 2024
June 2-6
Anaheim Convention Center
Anaheim, California
-
ASMS (American Society for Mass Spectrometry) 2024
June 2-6
Anaheim Convention Center
Anaheim, California