Read More
Nexera Series Featured Applications
Advanced Peak Deconvolution in HPLC using i-PDeA and the SPD-M40 Diode Array Detector

Baseline separation of chromatographic peaks can be difficult to achieve. Intelligent Peak Deconvolution Analysis II (i-PDeA II) extracts target peaks from partially co-eluting peak groups using the chemometrics multivariate curve resolution alternating least squares (MCR-ALS) technique with the SPD-M40 diode array detector. It is possible to obtain fast, accurate quantitative analysis for partially co-eluting peaks, as well as spectral identification. This lessens method development time and increases the confidence in the analytical results.
Impurity Analysis in Pharmaceutical Products with the Advanced Photodiode Array Detector SPD-M40

The following procedure is commonly used to measure the concentration of impurities in pharmaceutical products using area percentage normalization: prepare and analyze the standard solution at a concentration such that the height of the main peak will be within the linear range of the detector, then analyze multiple dilutions of the sample. Since the new photodiode array detector SPD-M40 has completely eliminated the effects of stray light during detection, it not only provides a wide dynamic range (linearity up to 2.5 AU as a specification value, typical value is more than 2.5 AU) but also achieves low noise and high sensitivity.
Improvement of Quantitative Performance for Ibuprofen Using UV Cut-Off Filter on SPD-M40
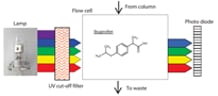
Ibuprofen is an example of a nonsteroidal anti-inflammatory drug (NSAID) and is widely used as an antipyretic or analgesic agent. It has been reported that during tests to confirm stability during storage, decomposition products were generated due to temperature, acidity, light irradiation, etc. of the surroundings. In particular, the area percentage of 4-isobutylacetophenone, one of the decomposition products, increased by about 40% after the 72-hour light irradiation test. The Nexera Series photodiode array detector SPD-M40 is equipped with a UV cut-off filter that exlcudes light in the ultraviolet range, in order to ensure more stable detection of compounds that are prone to photodegradation. We are introducing examples of improving the quantitation of ibuprofen using the UV cut-off filter function of the SPD-M40.
High-Resolution and High-Speed Simultaneous Analysis of Preservatives in Cosmetics Using SPP Column
This article introduces a method for the analysis of 24 types of cosmetic preservatives, either specified in the cosmetics standards established by the Japanese Ministry of Health, Labour and Welfare or regulated by the European Commission, using ultra-high performance liquid chromatography (UHPLC). Analyses were performed using the Nexera™ series UHPLC system equipped with a Shim- pack Velox™ C18 SPP (superficially porous particles, core-shell) column, which enabled both high resolution and reduced assay time.
High-Resolution and High-Speed Simultaneous Analysis of Regulated UV-Adsorbents in Cosmetics using SPP Column

Many cosmetics contain ultraviolet light absorbents (UV- absorbents) to protect the skin from ultraviolet light. In Japan, the standards for cosmetics based on the Pharmaceutical Affairs Law (Ministry of Health and Welfare Notice No. 331, 2000) regulate the types and allowable quantities of UV-absorbents that may be used in cosmetic products. Since regulations can vary significantly between different countries and regions, quantification with HPLC is used to determine compliance with import/export regulations. This report presents an example of high-throughput analysis of 23 kinds of UV-absorbents using the Nexera™ LC system and Shim-pack Velox™ C18 SPP (superficially porous particles, core-shell) column. This is a larger number of compounds than covered in a previous report as it also includes some residues regulated by EU agencies.
High-Speed Analysis of Linezolid following the Draft Guidance of International Harmonization of Pharmacopoeias

This article introduces an example of analysis of linezolid based on the USP and an example of its high-speed analysis based on the draft guidance of international harmonization of JP/USP/EP using Shimadzu Nexera™ Series and Shim-pack Scepter™ C18.
High-Speed Analysis of Methylated Catechin in Benifuuki Green Tea

It has been reported that "Food with Function Claims" Benifuuki green tea contains a large amount of methylated catechin as an antiallergic functional ingredient. (1) Methylated catechin is a kind of polyphenol which is contained in tea leaves, and is a partially methylated form of epigallocatechin gallate. It has also been reported that methylated catechin alleviates the discomfort of the eyes and nose caused by house dust and other forms of dust. This article introduces applications of analysis of methylated catechin in Benifuuki green tea leaves based on the JAS method and its high-speed analysis, using a Nexera™ X3 and Shim-pack™ GIST Series columns.
Simple and Quick Analysis of Theanine in Teaby Automatic Pre-Column Derivatization Method

Theanine, which has an umami taste, accounts for more than half of the free amino acids in tea and is an important component that determines the flavor of tea. It is known that there is a strong correlation between theanine content and tea quality, especially for green tea. Theanine also has various physiological effects, such as relaxing effect, stress-reducing effect, premenstrual syndrome (PMS) alleviation, and so on. With the recent health food boom, theanine is getting attention day by day. Application News L529A introduced amino acid analysis by the automatic pre-column derivatization method using the co-injection mode of the i-Series. This article introduces an example of simple and quick analysis of theanine and other major amino acids, by the same method as L529A using the Nexera™ Series.
Qualitative Analysis of UV-Absorbents in Cosmetics Based on UV-Vis Spectrum

Many cosmetic products contain ultraviolet absorbers (UV- absorbents) which protect the skin from ultraviolet rays. In Japan, ingredients of UV-absorbents and their allowable quantities are regulated under Standards for Cosmetics (2000, Ministry of Health, Labour and Welfare Notification No. 331) based on the Act on Securing Quality, Efficacy and Safety of Products Including Pharmaceuticals and Medical Devices (PMD Act). Because these standards differ depending on the country and region, compliance with the applicable regulations is verified by high performance liquid chromatography (HPLC) when importing/exporting cosmetics. Against this background, Application News No. L541 introduced an example of high-speed analysis of 23 UV-absorbents. This article introduces a method for qualitative analysis of six UV-absorbents based on the UV-Vis spectrum.
Monitoring Organic Acids during Fermentation with Shim-pack Fast-OA High-Speed Organic Acid Analytical Column
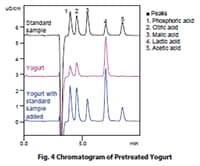
Using a Shim-pack Fast-OA column to analyze yogurt, we were able to confirm the primary organic acids in about 12 minutes. These results from rapid analysis of organic acids suggest that the method provides and effective means of ensuring an adequate number of repeated analyses for multianalyte processing and quickly controlling the fermentation process based on the fermentation status.
Fermentation Processes Monitoring Using a Nexera™ Dual Injection System
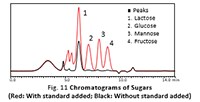
When microorganisms decompose substances to produce useful materials, that process is called fermentation. Fermentation is used not only for producing foods, but in recent years is also being widely used in industrial fields. In such fields, organic acids, sugars, amino acids, and other groups of compounds are multilaterally measured to understand the fermentation process or optimize conditions. In the case of HPLC analysis, the appropriate separate mode and detection method can differ depending on the component class. This requires multiple independent pieces of equipment for each analysis. This article introduces an example of monitoring a fermentation process using a dual injection system to evaluate two types of analysis simultaneously with only one system.
Analysis of Meloxicam in Accordance with the United States Pharmacopoeia by Nexera XR

Meloxicam, inhibits the biosynthesis of prostaglandin, is utilized as an anti-inflammatory and analgesic for arthritis. In the United States Pharmacopoeia (USP), the HPLC method using a UV-Vis absorbance detector is adopted as the test method for meloxicam. This article introduces an example of the analysis of meloxicam in accordance with the USP by Nexera XR, and compatibility with ProminenceTM series using ACTO (Analytical Condition Transfer and Optimization) function.
Quick Estimation of the Freshness and the Level of Putrefaction in Fish Meat Using Nexera™ Dual Injection System

This article introduces simultaneous determination of the K value as an indicator of freshness and histamine as an indicator of the level of putrefaction using the Nexera dual injection system. Under the analytical conditions described in this article, amino acids and nucleic acids known as nutritional compounds, including umami taste, contained in fish meat were able to be determined simultaneously as well.
Improvement of Productivity in Research on Intestinal Microbiota by Shim-pack™ Fast-OA High-Speed Organic Acid Analysis Column
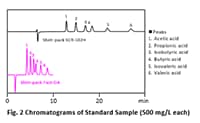
Short-chain fatty acids, which are an object of research in the field of intestinal microbiota, were analyzed by using a Shim- pack Fast-OA column. Quantitation of the target compounds was possible within a time of 10 min and was unaffected by contamination. As a result of measurements of the amounts of short-chain fatty acids contained in feces samples from 5 monkeys, virtually no short-chain fatty acids were found in one sample that was collected and frozen more than half a day after excretion, but it was possible to measure the existence of short-chain fatty acids with satisfactory sensitivity in the samples that were frozen immediately after excretion.
USP-Compliant Analysis of Vitamins in Dietary Supplements Analysis of Calcium Pantothenate by Nexera™ XR
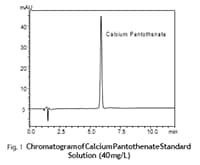
The United States Pharmacopeia (USP) provides standards for quality control of dietary supplements and specifies testing methods and judgment standards for dietary supplements. Because many dietary supplements are distributed globally, USP compliance has become an important judgment standard for verifying the quality of supplements for consumers. This article introduces an example in which the calcium pantothenate in a dietary supplement was analyzed using a Nexera XR, which is part of the Shimadzu Nexera Series of ultra high performance liquid chromatographs.
Analysis of Formaldehyde Using HPLC and Post-Column Derivatization with Acetylacetone

Formaldehyde is a useful ingredient for wood preservatives and resin products, but because it is a causative agent of sick house syndrome, it has attracted attention. Shampoos, skin lotion, and foundation products used in everyday life are generally called cosmetics, and the ingredients used in them are subject to particularly strict regulation because they are applied to the human body. Japan’s Standards for Cosmetics (Ministry of Health and Welfare Notification No. 331, 2000) list formaldehyde as one material which is prohibited to include in cosmetics. Moreover, in the EU, the content of formaldehyde in nail polish and other nail products is limited to no more than 5%. In this article, formaldehyde in cosmetics was detected using the HPLC method and the post-column derivatization with acetylacetone, which is an established test method under the Methods of Analysis in Health Science (The Pharmaceutical Society of Japan, 2015). This article introduces examples of analysis of formaldehyde using a Nexera™ Series Nexera XR ultra high performance liquid chromatograph.



