LC/MS/MS Method Package for Short Chain Fatty Acids
For LabSolutions™ LCMS Software
Short-Chain Fatty Acids and Organic Acids Targeted for Analysis

Of the short-chain fatty acids produced by intestinal bacteria, acetic acid, propionic acid, and butyric acid are well known, and it has been reported that there are some connections between them and lifestyle-related diseases such as obesity and diabetes. Generally speaking, short-chain fatty acids are highly volatile and highly hydrophilic. This makes it difficult to perform LCMS analysis using a conventional reverse phase system. For that reason, this method package targets short-chain fatty acids (C2 to C5) that have been derivatized using 3-nitrophenylhydrazine (3-NPH). After setting MRM transitions, it can be used for the simultaneous analysis (of 22 components), including organic acids related to the central metabolic pathways.
|
|
MRM Transition Settings
When making MRM transition settings, selectivity can be enhanced by specifying characteristic product ions for 3-nitrophenylhydrazine derivatives. Note that some organic acids have a carbonyl group derived from ketones, so 3-nitrophenylhydrazine derivatives that reacted with carboxylic acid and the carbonyl group are targeted for MRM transition.
Pretreatment Protocol Including Derivatization
The entire protocol necessary for pretreatment, including the derivatization step using 3-nitrophenylhydrazine, is contained in the Instruction Manual for this method package. By following these procedures, an analyst should be able to perform the entire process, from derivatization to data acquisition and data analysis, immediately after the package is introduced.
For the derivatization using 3-nitrophenylhydrazine, carbodiimide is used as a condensing agent, and reaction proceeds under mild conditions. Moreover, in order to efficiently carry out reactions with both short-chain fatty acids and organic acids, this method package includes derivatization using 3-NPH instead of the more commonly used 2-NPH. Also, 3-NPH derivatizations after reactions are stable, and quantitative analysis of the short-chain fatty acids or organic acids in the sample can be performed through reaction with an appropriate amount of derivatization reagent. Additionally, as reactions take place in water solutions using this derivatization reagent, the sample will not be dried and volatile components will not be lost.
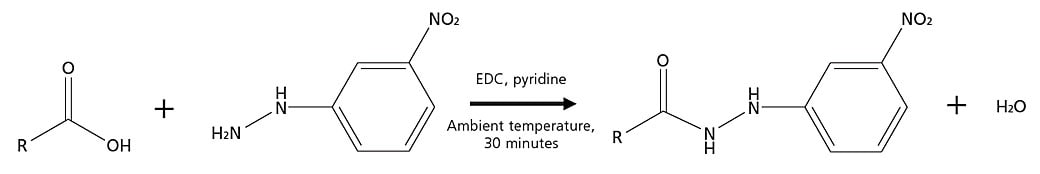
Derivatization Reaction with 3-nitrophenylhydrazine (3-NPH)
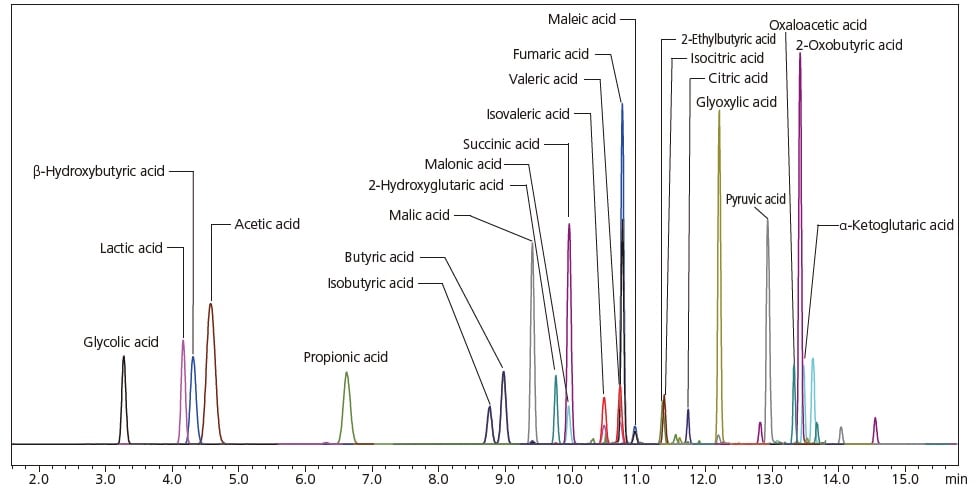
The following example shows an analysis using the LCMS-8060 of a standard mixed solution of short-chain fatty acids/organic acids. An MRM chromatogram of short-chain fatty acids/organic acids derivatized using 3-NPH is shown. As can be seen, sufficient separations of the components, such as the structural isomers butyric acid, isobutyric acid, fumaric acid, maleic acid, citric acid, and isocitric acid were obtained. This method package includes MRM transitions that were formulated beforehand in order to achieve optimization for 3-NPH derivatizations for short-chain fatty acids/organic acids. As a result, short-chain fatty acids (C2 to C5) and organic acids representative of central metabolic pathways, which are difficult to analyze with LCMS using a conventional reversed phase system, can be analyzed simultaneously.
Remarks and Precautions
LabSolutions LCMS Ver. 5.99SP2 or later is required.
LabSolutions is a trademark of Shimadzu Corporation.
News / Events
-
MSACL (Mass Spectrometry Applications to the Clinical Lab) 2025
September 23-25
Hotel Bonaventure Conference Center
Montreal, Canada
-
Shimadzu Announces New LCMS-8065XE Triple Quadrupole Mass Spectrometer
Shimadzu Scientific Instruments announces the LCMS-8065XE, the latest evolution in triple quadrupole mass spectrometry that redefines analytical excellence with world-class sensitivity, efficiency and sustainability.
-
Shimadzu Announces New LCMS RX Triple Quadrupole Mass Spectrometry Series
Shimadzu Scientific Instruments announces the new LCMS RX series of triple quadrupole mass spectrometry instruments. These advanced systems are supported by integrated hardware and software technologies to ensure reliable and robust results at lower operating costs, even under the most challenging and dynamic laboratory environments.
-
Innovative Technology Developed by Shimadzu and Washington University is Expected to Improve Healthy Life Expectancy
-
New Q-TOF LCMS-9050 Delivers Exceptional Mass Accuracy and Fast, Stable Polarity Switching
-
Achieve Effortless High-Speed High-Sensitivity Analysis With New Space-Saving LCMS-2050 Single Quadrupole Mass Spectrometer
Shimadzu Scientific Instruments introduces the LCMS-2050 Liquid Chromatograph Quadrupole Mass Spectrometer that features a significantly reduced size, while providing high speed and high sensitivity analysis. Designed for effortless operation, the LCMS-2050 mass spectrometer combines ease of use as an LC detector with excellent MS capabilities.




