The Column
6.1. Gas Chromatography Columns
Two types of columns are used in gas chromatography: packed columns and capillary columns.

Short, thick columns made of glass or stainless steel tubes, packed columns have been used since the early stages of gas chromatography.
Packed columns produce broad peak shapes and have low separation performance, but can also handle large sample volumes and are not susceptible to contamination. They are still used today in official analytical methods and for gas analysis.

Currently the prevailing column type, capillary columns produce sharp peak shapes, achieve excellent separation performance, and are suited to high-sensitivity analysis.
Viewing a cross-section image of a packed column reveals a tube filled with a particulate substance called packing. Packed columns have been used throughout the long history of gas chromatography, and many different packed columns have been created for different analytical applications. In contrast, typical capillary columns consist of a thin, fused silica glass tube with a thin, internal liquid phase coating. Capillary columns were developed after packed columns, and though there are fewer types of capillary columns, their separation performance is dramatically superior to packed columns.
Packed Column
Stainless steel or glass tube filled with particulate packing material (an adsorbent material, or a support material coated or impregnated with a solid phase).
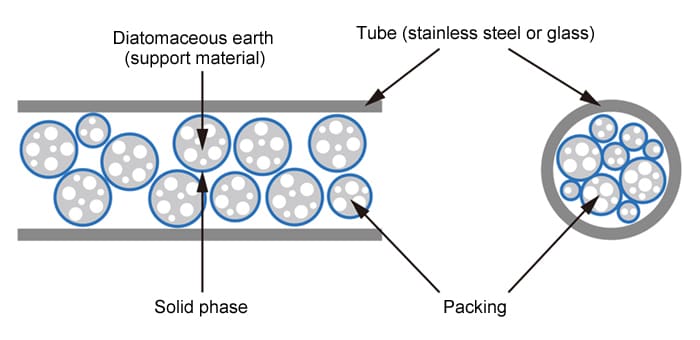
- Internal Diameter: 2 to 4 mm
- Length: 0.5 to 5 m (most commonly 2 m)
- Packing: Support material with 0.5 to 25 % liquid phase (partition material) or no liquid phase (adsorbent material)
- Liquid Phase: Multiple types available
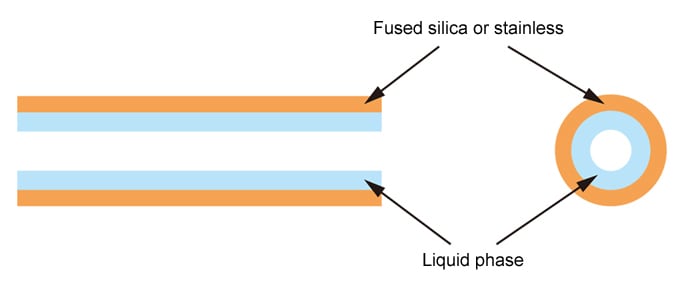
Capillary Column
A typical capillary column is a thin, fused silica glass tube, lined with a liquid phase or adsorbent material or having a chemical bonding layer. Thin metal tubes are also sometimes used as capillary columns.
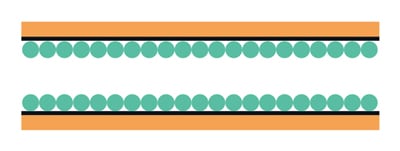
PLOT column
(contains immobilized porous polymer/alumina, etc.)
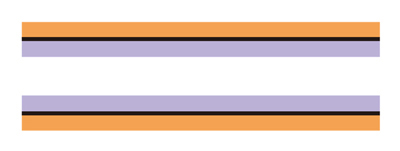
WCOT or chemical bonding column
(lined with liquid phase or a chemical bonding layer)
- Internal Diameter: 0.1, 0.25, 0.32, 0.53 mm
- Length: 5 to 100 m (most commonly 30 m)
- Material: Fused silica glass
- Liquid Phase: Good separation but less variety than packed columns
6.2. Column Type and Effect on Separation
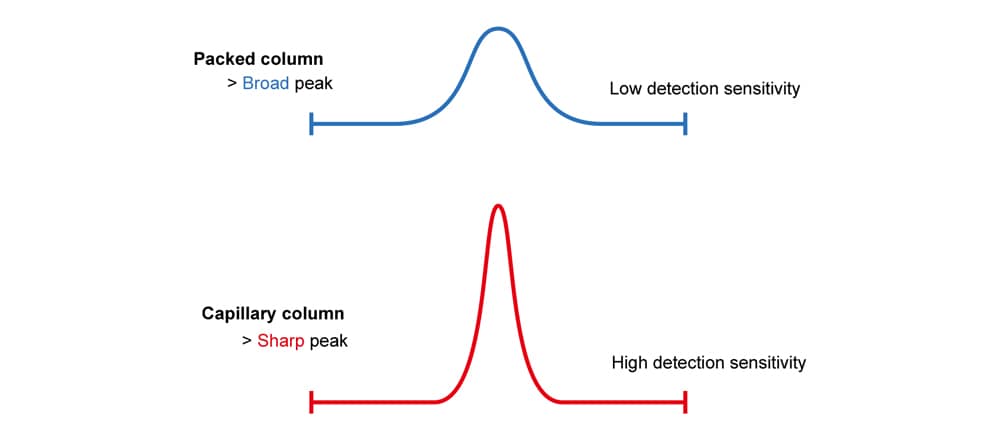
Packed columns produce broad peaks and capillary columns produce sharp peaks.
In addition, capillary columns produce taller peaks, which allows the detection of lower concentrations (high detection sensitivity). This is the advantage of capillary columns.
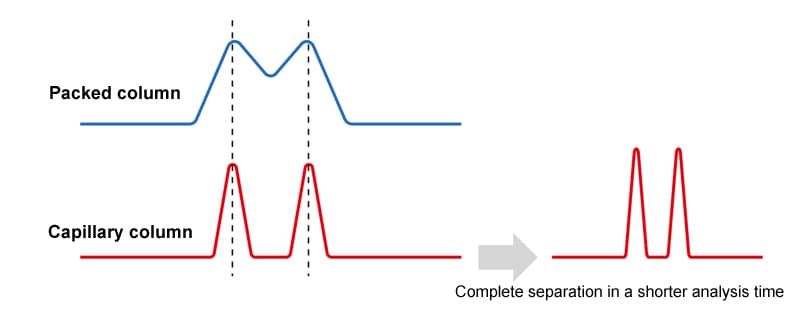
Sharper peaks provide better separation but also shorter analysis times.
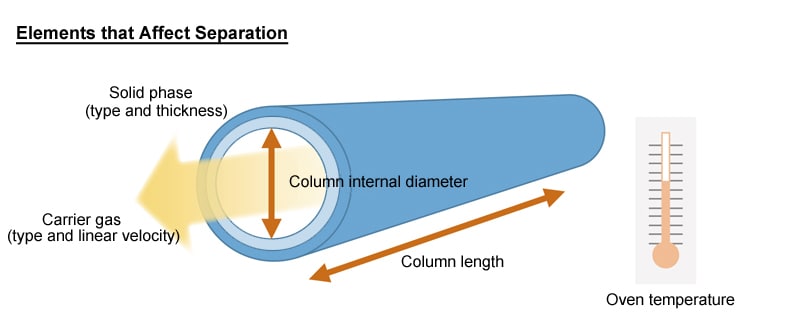
6.3. General Guide to Column Selection
Component separation is affected by the following elements.
Classification of Capillary Column Liquid Phases
| Type of Solid Phase | Polarity | Separation Characteristics | Application | Operational Temperature Range (Approx.) |
|---|---|---|---|---|
| Methyl silicone | Non-polar | Boiling point order | Petroleum, solvents, high boiling point compounds | -60 to 360 °C |
| Phenylmethyl | Slightly polar | Moderately polar |
Phenyl groups retain aromatic compounds. | Perfumes, environmental compounds, aromatic compounds | -60 to 340 °C |
| Cyanopropyl phenol | Moderately polar | Strongly polar |
Effective at separating oxygen-containing compounds, isomers, etc. | Agricultural chemicals, PCBs, oxygen-containing compounds *Better to avoid use with FTDs (NPDs) |
-20 to 280 °C |
| Trifluoropropyl | Moderately polar | Strongly polar |
Specifically retains compounds that contain halogens. | Halogen-containing compounds, polar compounds, solvents | -20 to 340 °C |
| Polyethylene glycol | Strongly polar | Strong retention of polar compounds | Polar compounds, solvents, perfumes, fatty acid methyl esters | 40 to 250 °C |
General Guide to Selecting Polarity
- Selecting columns with polar properties that are close to the polarity of the target compounds
- Analysis of non-polar compounds → Non-polar column
- Analysis of polar compounds → Strongly polar column
- Selection by analytical objective
- Large difference in boiling point between analytical target compounds → Non-polar column
- Isomers or other compounds with little difference in boiling points → Strongly polar column
Guide to Selection of Internal Diameter, Length, and Coating Thicknes
- Selection based on required separation
- High-resolution separation required → Internal diameter: Thin, Length: Long
- Adequate separation with shorter analysis time → Internal diameter: Thick, Length: Short, Coating thickness: Thin
- Selection by analytical objective
- Analysis of low boiling point compounds → Length: Long, Coating thickness: Thick
- Analysis of high boiling point compounds → Length: Short, Coating thickness: Thin


